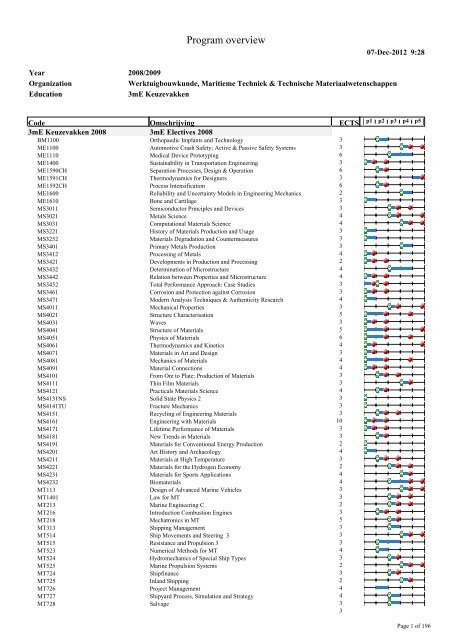
Section 6 3 Periodic Trends Study Guide Answers

A high ionization energy value indicates that the atom has a strong hold on its electrons and is not likely to lose an outer electron and form a positive ion.
Section 6 3 periodic trends study guide answers. Section 6 3 periodic trends in your textbook read about atomic radius and ionic radius. Identify the four blocks of the periodic table based on electron. The atomic radius within these groups increases as the atomic number increases.
Look at the data for the alkali metals and noble gases. Circle the letter of the choice that best completes the statement or answers the question. Section 6 3 periodic trends 171 group trends in atomic size in the figure 6 14 graph atomic radius is plotted versus atomic number.
Ionization energy is the energy required to remove an electron from a gaseous atom. Chapter 6 periodic table trends study guide name if you can answer these questions you should be able to earn a 100 on the quest tomorrow. Circle the letter of the choice that best completes the statement or answers the question.
Explain why elements in the same group have similar properties. Atomic radii cannot be measured directly because the electron cloud surrounding the nucleus does not have a clearly defined c. Trace the development and identify key features of the periodic table.
Part a completion learn with flashcards games and more for free. This increase is an example of a trend. 1 7 the period signifies the number of energy levels an element has.
Section 6 3 periodic trends 1. Periodic table and trends. Prentice hall chemistry 2005 learn with flashcards games and more for free.